In vitro wound-healing assay also known as the scratch assay
The wound-healing assay, also known as the scratch assay, is an established two-dimensional (2D) technique that can be used to investigate collective migration and wound healing in vitro [1], [2]. This method was one of the first to be developed for the study of cell migration and measures the rate at which cells, in a cell monolayer, migrate to fill a cell-free gap [1], [3]. Three-dimensional (3D) wound healing assays are beyond the scope of this article and have been succinctly covered by Stamm et al. 2016 [2].
------------------------------------------------------
Researchers can perform wound healing experiments with these CytoSMART devices: full-plate scanner Omni, fluorescence cell imager Lux3 FL, mini live-cell imagers Lux2 and Lux3 BR.
------------------------------------------------------
The wound healing assay is a simple and inexpensive method. While this assay does not recapitulate the exact conditions of a wound, it can be successfully applied to model and investigate cell movement in a controlled in vitro environment. The technique reproduces wounding by creating a gap in a confluent cell monolayer and consists of four main steps that will be elaborated upon below (see Figure 1 for an overview) [1].
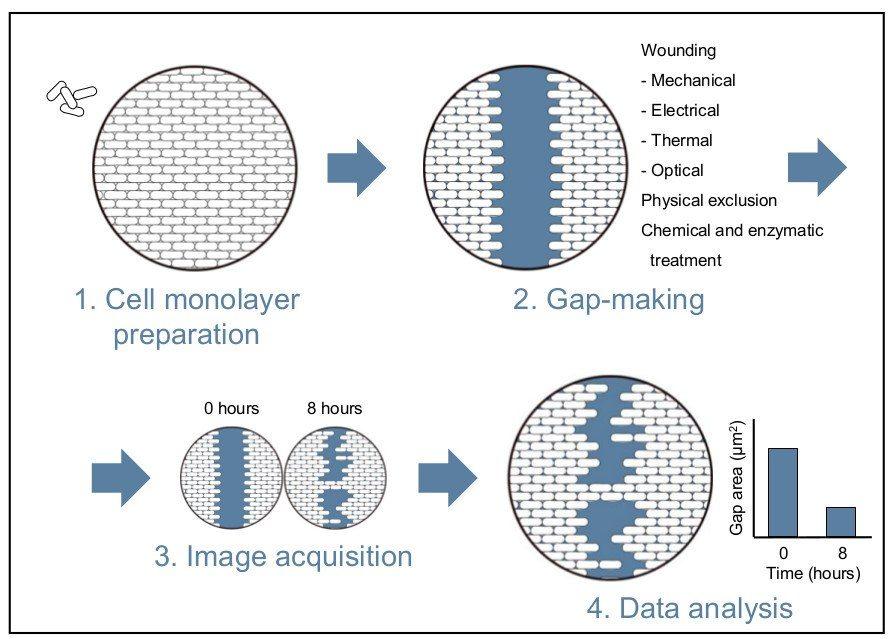
Wound healing assay protocol
Culture preparation
The first step of the assay is to culture a confluent cell monolayer. This monolayer represents the in vivo conditions of the tissue before wounding such as, an intact epithelium. Most often, epithelial and endothelial cells are used to produce the monolayer, and in particular, cell types capable of sheet migration [4]. Studies have also examined the migration of vascular smooth muscle cells [5], [6].
Depending on the cell type used in the assay, cell proliferation can be a confounding factor. Cell proliferation can compete with cell migration to fill the gap made during the assay. If this occurs, the cell medium can be optimized to reduce cell proliferation. Decreasing the concentration of serum (serum starvation) is the most common change. Proliferation inhibitors, such as mitomycin C, can also be added to the medium. Careful optimization is necessary as any changes to the medium can result in unpredictable time- and cell-type-dependent effects that can interfere in the assay [1].
Scratch-making
After the cells have become confluent, the next step is to make a cell-free gap in the monolayer. The most frequently used method is to wound the monolayer by mechanical scratching (scratch wound) or stamping. Alternatives to mechanical damage can include thermal, electrical, and optical wounding [2], [7]. In microfluidic wound healing assays, chemical and enzymatic treatments have been used to clear cells from fluid channels [5], [6], [8], [9]. Methods that avoid cell damage altogether, such as physical exclusion, have also been described [4], [10].
The preparation of gaps can be performed manually or be automated. Drawbacks with the manual method of wounding include low throughput and well-to-well variation of the gap width [1], [11]. This reproducibility can be improved with the use of commercial tools for making uniform gaps [1]. Modifications to the wound healing assay that make use of automation can increase both throughput and improve reproducibility [2], [7]. Ensuring reproducibility is important for the following data acquisition step.
Data acquisition – scratch images for time-lapse
With a cell-free gap prepared, optical microscopy can be used to observe cells migrating into the wound area. Cell migration is best viewed using phase-contrast imaging rather than fluorescence, and the wound area in the field-of-view should be maximized using the objective lens [12], [13].
Once the microscope is set up, a series of time-lapse images (snapshot method) can be acquired as cells migrate into the cell-free gap [1]. These time points should be collected within 24 hours of the experiment being started to minimize the confounding effects of cell replication on gap closure. The migration images can then be used to collect measurements or be visually scored [4]. Accurate measurements can be manually acquired using a digital camera fitted to the microscope; however, this process is time-consuming, and maintaining the same field-of-view along each gap can be difficult [4].
The disadvantages experienced with manual acquisition can largely be overcome using automation that allows for live-cell imaging. Aspects of acquisition that can be automated include image capturing, point visiting, and environmental control [2], [4]. Automation also has greater functionality; for example, it can be used to determine the experimental endpoints and generate kinetic, functional, and quantitative measurements of living cells (see Figure 2) [1].
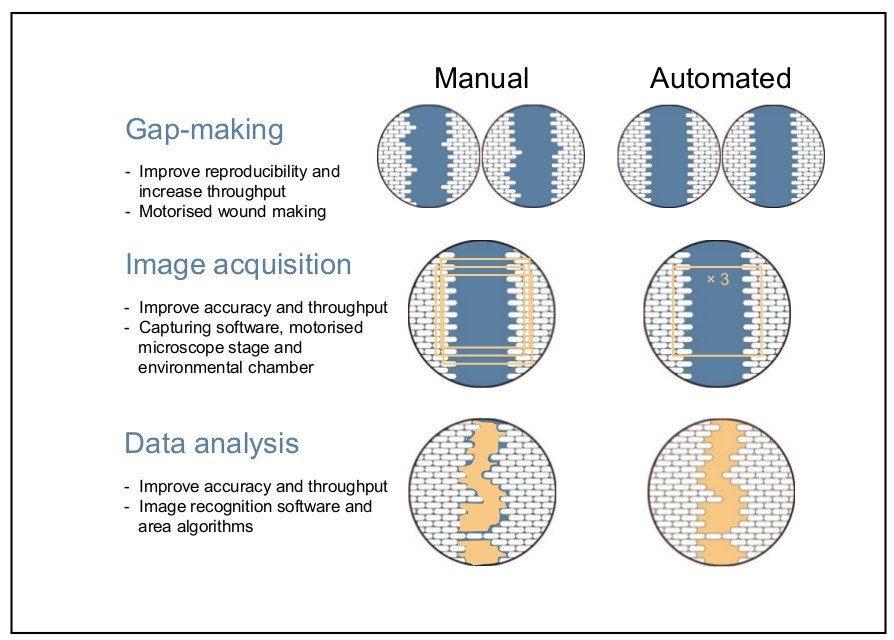
--------------
Whole-well imaging systems can be applied to the wound-healing assay. For researchers interested in such a functionality see the CytomSMART Omni landing page. This live-cell imaging system operates from within cell culture incubators and is fully automated, to easily create time-lapse videos of gap closure.
---------------
Data analysis - scratch closure
Once images of the gap closing have been acquired, several methods of analysis can be used to quantify the rate of cell migration. The first method measures the change in the wound width (nanometres) over time. This width is the average distance between the two margins of the scratch. The second method calculates the change in wound area over time as a percentage of wound closure. These two methods can be time-consuming when performed manually. The final method measures the relative wound density over time expressed as a percentage and is most often applied in live-cell imaging software [1], [12]. This method calculates the ratio of the occupied area in the gap to the total area of the initial gap. The relative wound density method can be further refined to factor in cell proliferation or pharmacological effects. This can be achieved by counting cells in subregions found inside and outside the wound area to determine relative cell density [12]. Total cell count within the wound area can also be used to assess cell migration and wound healing [2].
In addition to measuring changes in the wound area, the wound healing assay can also be applied to tracking the movement of individual cells at the leading edge of the wound. This allows researchers the opportunity to understand the role of genes in the regulation of cell migration [3].
Most wound healing assays still use manual data acquisition and analysis in part. The manual extraction of data is very time-consuming, subjective, and prone to error. Therefore, the analysis of large datasets remains a bottleneck with many assays. In addition, the multitude of assays available makes comparison between experiments difficult [2]. Automated data analysis software, such as TScratch and ImageJ, has been developed that can significantly accelerate analysis and overcome limitations associated with image quality [14], [15].
For in-depth wound healing assay guidelines on parameters to ensure quantitative and reproducible results, please see the review by Jonkman et al. (2014) [4].
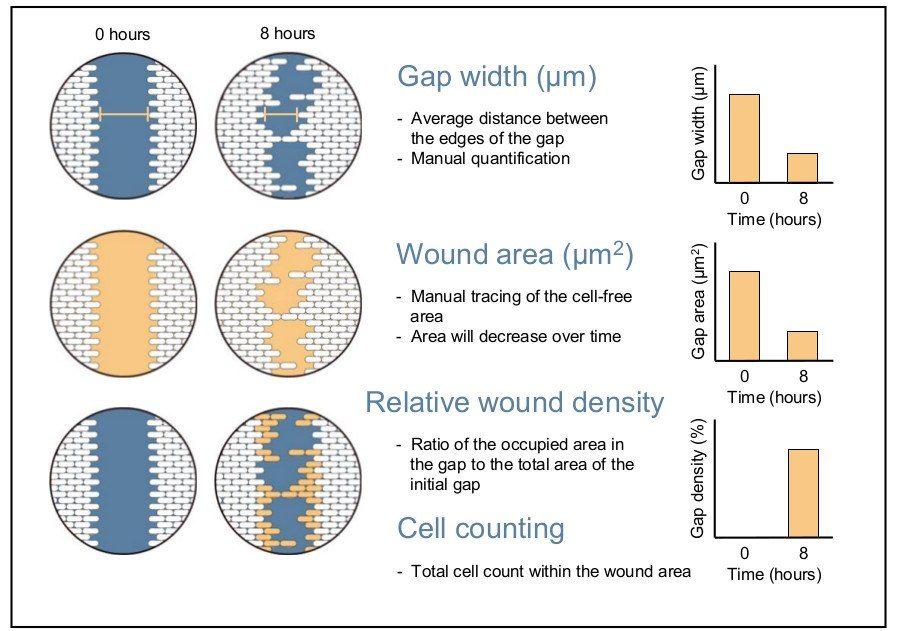
Cell migration assay
The wound healing assay is used to investigate cell migration and wound healing. Cell migration is the movement of individual cells, cell sheets, and clusters from one location to another, and two main types have been identified, namely single-cell migration and collective cell migration. The latter is defined by Grada et al. (2016) as the coordinated movement of a group of cells that maintains their intercellular connections and collective polarity [1].
Collective migration can take on two different forms depending on the extracellular matrix. Three-dimensional collective migration takes place on a tissue scaffold, and it is the movement of cells organized in a multicellular network of strands. Two-dimensional (2D) collective migration (sheet migration) takes place across a tissue surface, and it is the movement of flat monolayer sheets. Wound healing is an example of sheet migration [1].
-----------
For researchers looking to evaluate single-cell migration patterns, we recommend the CytoSMART Lux2 Duo Kit. This mini-live cell imaging system operates from within cell culture incubators and allows for side-by-side comparison between cell cultures.
-----------
Wound healing
Four processes occur during wound healing, namely haemostasis, inflammation, migration-proliferation, and maturation-remodeling [1], [2]. Upon injury and under the influence of growth factors and cytokines, keratinocytes at the rear of the wound margin begin to proliferate and migrate onto the wound bed. This process involves cell migration, proliferation, and differentiation [1].
Migration is considered the rate-limiting process during healing, and therefore migration assays are a key part of investigating wound healing [2].
Applications of the wound healing assay
The wound healing assay is a convenient and economical method to investigate collective cell migration under different experimental conditions. As collective cell migration is linked to many physiological and pathological processes related to embryogenesis, wound repair, and cancer metastasis, the wound healing assay is broadly applicable [4], [8]. The assay can be used to investigate the effects of the cell-matrix and cell-cell interaction on cell migration and be combined with transfection to determine the effect of the expression of exogenous genes on the migration of individual cells [1], [3].
The assay is also scalable allowing for high-throughput screening of genes involved in cancer cell migration, small molecule discovery, and drug discovery [1], [2], [16]. Examples of some of these applications are presented below.
A microfluidic wound healing assay was developed by Wei et al. (2015) to investigate vascular smooth muscle cell (VSMC) migration. The migration of these cells after endothelial injury is an inherent factor in the progression of atherosclerosis and the complications associated with intimal hyperplasia [6]. Both of these diseases are a major cause of morbidity and mortality globally [17]. Understanding the processes involved in this migration can provide potential targets for inhibition. This modified wound healing assay is a closer representation of the microenvironment present in the vasculature. In the study microfluidic wound healing assay uses five types of VSMCs [6].
The wound healing assay can be used in the study of wound dressings. Alves et al. (2020) used the assay to investigate novel hydrogel blends as a potential wound dressing. The effect of the hydrogel on wound closure was assessed by incorporating it into the cell medium. The authors demonstrated that hydrogels could be used to improve the wound healing process by promoting fibroblast migration, adhesion, and proliferation [18].
Inhibitors of cell invasion and metastasis can be effectively screened using the wound healing assay. Wang et al. successfully screened cytotoxic alkaloids for the ability to inhibit biological processes related to cell migration and cytoskeleton dynamics. The study was able to identify certain cytotoxic alkaloids as anti-migrating agents that could be investigated further [19].
------------------------------------------------------
References
[1] A. Grada, M. Otero-Vinas, F. Prieto-Castrillo, Z. Obagi, and V. Falanga, “Research techniques made simple: analysis of collective cell migration using the wound healing assay,” J. Invest. Dermatol., vol. 137, no. 2, pp. e11--e16, 2017.
[2] A. Stamm, K. Reimers, S. Strauß, P. Vogt, T. Scheper, and I. Pepelanova, “In vitro wound healing assays--state of the art,” BioNanoMaterials, vol. 17, no. 1–2, pp. 79–87, 2016.
[3] L. G. Rodriguez, X. Wu, and J.-L. Guan, “Wound-healing assay,” in Cell Migration, Springer, 2005, pp. 23–29.
[4] J. E. N. Jonkman et al., “An introduction to the wound healing assay using live-cell microscopy,” Cell Adh. Migr., vol. 8, no. 5, pp. 440–451, 2014.
[5] A. D. der Meer, K. Vermeul, A. A. Poot, J. Feijen, and I. Vermes, “A microfluidic wound-healing assay for quantifying endothelial cell migration,” Am. J. Physiol. Circ. Physiol., vol. 298, no. 2, pp. H719--H725, 2010.
[6] Y. Wei et al., “A tubing-free microfluidic wound healing assay enabling the quantification of vascular smooth muscle cell migration,” Sci. Rep., vol. 5, p. 14049, 2015.
[7] C. R. Keese, J. Wegener, S. R. Walker, and I. Giaever, “Electrical wound-healing assay for cells in vitro,” Proc. Natl. Acad. Sci., vol. 101, no. 6, pp. 1554–1559, 2004.
[8] R. Riahi, Y. Yang, D. D. Zhang, and P. K. Wong, “Advances in wound-healing assays for probing collective cell migration,” J. Lab. Autom., vol. 17, no. 1, pp. 59–65, 2012.
[9] J.-Y. Lin, K.-Y. Lo, and Y.-S. Sun, “A microfluidics-based wound-healing assay for studying the effects of shear stresses, wound widths, and chemicals on the wound-healing process,” Sci. Rep., vol. 9, no. 1, pp. 1–11, 2019.
[10] A. P. Looney and M. Bhattacharya, “Fibroblast Gap-closure Assay-Microscopy-based in vitro Assay Measuring the Migration of Murine Fibroblasts,” Bio-protocol, vol. 9, no. 16, 2019.
[11] S. Martinotti and E. Ranzato, “Scratch Wound Healing Assay,” 2019.
[12] S. T. Johnston, E. T. Shah, L. K. Chopin, D. L. S. McElwain, and M. J. Simpson, “Estimating cell diffusivity and cell proliferation rate by interpreting IncuCyte ZOOMTM assay data using the Fisher-Kolmogorov model,” BMC Syst. Biol., vol. 9, no. 1, p. 38, 2015.
[13] C. N. Ramirez et al., “Validation of a high-content screening assay using whole-well imaging of transformed phenotypes,” Assay Drug Dev. Technol., vol. 9, no. 3, pp. 247–261, 2011.
[14] T. Gebäck, M. M. P. Schulz, P. Koumoutsakos, and M. Detmar, “TScratch: a novel and simple software tool for automated analysis of monolayer wound healing assays: Short Technical Reports,” Biotechniques, vol. 46, no. 4, pp. 265–274, 2009.
[15] K. A. Main, C. M. Mikelis, and C. L. Doçi, “In Vitro Wound Healing Assays to Investigate Epidermal Migration,” 2019.
[16] J. C. Yarrow, Z. E. Perlman, N. J. Westwood, and T. J. Mitchison, “A high-throughput cell migration assay using scratch wound healing, a comparison of image-based readout methods,” BMC Biotechnol., vol. 4, no. 1, p. 21, 2004.
[17] K. Kobiyama, R. Saigusa, and K. Ley, “Vaccination against atherosclerosis,” Curr. Opin. Immunol., vol. 59, pp. 15–24, 2019.
[18] A. Alves et al., “Xanthan Gum--Konjac Glucomannan Blend Hydrogel for Wound Healing,” Polymers (Basel)., vol. 12, no. 1, p. 99, 2020.
[19] X. Wang, C. C. Decker, L. Zechner, S. Krstin, and M. Wink, “In vitro wound healing of tumor cells: inhibition of cell migration by selected cytotoxic alkaloids,” BMC Pharmacol. Toxicol., vol. 20, no. 1, pp. 1–12, 2019.

