What are spheroids?
For decades, isolated cells grown in 2D monolayers on plastic or glass have been used to study a range of biological processes. While these culturing conditions are easy to control and are conducive to a variety of analytical techniques, they do not represent the complex 3D environments that cells are actually exposed to in vivo.
A number of 3D cell culturing methods have been developed that more accurately recapitulate cells’ native environments1. During embryonic development, cells aggregate and self-assemble into complex multi-cellular structures2.
The same aggregation process can likewise occur in in vitro culture systems if cell-cell cohesion is favored over cell attachment to a substrate2. The resulting cell aggregates are known as spheroids2.
Get insights into the spheroid formation, translucency, diameter, and growth kinetics with these CytoSMART devices: full-plate scanner Omni, fluorescence cell imager Lux3 FL, mini live-cell imagers Lux2 and Lux3 BR.
Spheroids are dense, 3D aggregates of cells that exhibit extensive cell-cell adhesion, typically retain their endogenous extracellular matrix, and have properties that closely mimic their in vivo tissue counterparts. Although scientists have been culturing cell aggregates for decades3,4, it was not until 1971 that the word “spheroid” was first used5. 3D cell culture was further popularized through the work of Mina Bissell in the 1980s6.
Since then, spheroids have become an exciting cell culture model, as they better mimic in vivo tissue properties and exhibit a superior capacity for differentiation compared to 2D culture systems1. These properties make them well-suited for use in drug development and tissue engineering studies, as well as for the study of fundamental biological principles.
Content
- Properties of spheroids
- Label-free microscopy to assess spheroid properties
- Evaluating spheroid quality
- Culturing methods
- Hanging drop
- Liquid overlay
- Pellet culture
- Spinner culture
- Biomaterials
- Culture conditions
- Tumor spheroids: example protocol
- Applications of spheroid culture
- Tumor spheroids
- Tissue engineering and repair
- Basic biology research
- Conclusion
Properties of spheroids
Spheroidal cultures exhibit greater physiological relevance than 2D cell cultures, including improved cell viability, morphology, proliferation, differentiation, metabolic activity, migration, angiogenesis, and gene expression12.
For example, spheroids composed of multipotent mesenchymal stem cells can differentiate more efficiently into chondrogenic, adipogenic, neurogenic, osteogenic, and hepatogenic lineages than these cells do in 2D culture2,13,14.
Spheroids may consist of one cell type, or incorporate multiple different cell types into a single co-culture spheroid2. Multicellular co-culture spheroids have been shown to be particularly successful in recapitulating the morphology and polarity of cell-cell adhesions that occur in vivo2. For example, artificial pancreatic islet spheroids that mimicked the morphology and hormone secretion activity of isolated naïve pancreatic islets were created by co-culturing three different pancreatic cell types together11.
Spheroid size is dependent on the cell type(s) from which they are formed and their culture conditions. They typically range from ~100 μm to >500 μm7,8. For example, adipose stem cells seeded in the presence of a biocompatible scaffold form spheroids of 80 – 110μm diameter9, while some cancer cell-derived tumor spheroids may reach diameters of over 1000 μm8. However, as their oxygenation is dependent on diffusion, spheroids larger than a few hundred micrometers exhibit upregulation of hypoxia-induced survival factors, including HIF-1α, and a loss of cell viability at their cores15,16.
This characteristic of spheroids has proven useful for the study of solid tumors, as larger tumor spheroids (~500 μm diameter) form nutritional and oxygen gradients that mimic their native counterparts10 (Figure 1). Tumor spheroids typically consist of a proliferating ring of cells that surrounds a quiescent interior7, and may additionally form a pathophysiological necrotic core10.
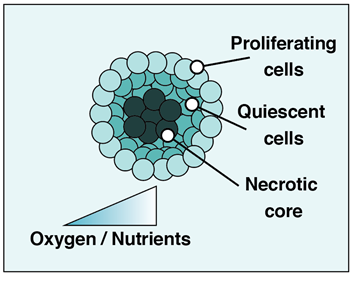
Label-free microscopy to assess spheroid properties
Spheroid morphology can be visually assessed using label-free brightfield or phase-contrast microscopy, which is low cost, easy to use, and prevents photodamage to the spheroids.
The simplest way to assess the properties of spheroids is to perform imaging with a small microscope that can sit inside the incubator with the spheroid culture plate. These microscopes can image the entire surface of a well and automatically stitch the images together, allowing spheroid formation to be easily followed without disruption.
A number of morphological parameters can be tracked over the course of spheroid formation in a semi-automated way using this technology (Figure 2). Spheroid integrity, volume, diameter, external morphology, translucency (an indicator of spheroid compaction), and growth kinetics (including growth delay) can all be assessed1. Further, the invasive potential of tumor spheroids can be measured by tracking cell migration into Matrigel28. All of these assays can be used in drug screening, drug response, and toxicology assays8.
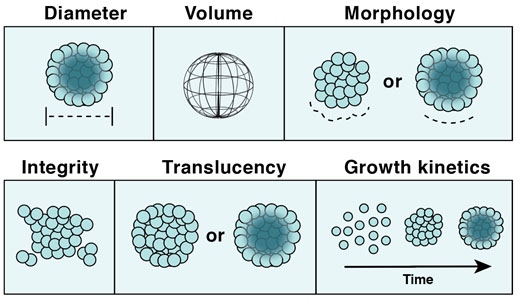
Evaluating spheroid quality
Using brightfield microscopy, quality spheroids appear as translucent balls with a well-defined exterior boundary and a slightly darker core21. Irregularly shaped or very loose cell aggregates represent spheroids of poor quality. In addition, if spheroid formation is allowed to progress too long, they may become darker over time, which corresponds to increased cell compaction21. Further, cells at the spheroid boundary may begin to dissociate from the main body21.
Spheroid quality can also be assessed using cell-labeling techniques. For example, while cell aggregates will stain homogenously with a membrane-specific dye, more compact spheroids will exclude dye from the interior21. In addition, vital dyes can be used to assess the viability of spheroids.
Culturing methods
Spheroid formation is dependent on the tendency of cells in suspension to aggregate and self-assemble into multi-cellular groupings17. This process likely involves the sequential induction of β1-integrin-dependent adhesion which facilitates cell aggregation, followed by strong homophilic E-cadherin-dependent cell-cell adhesion that drives spheroid compaction18,19. There are a number of distinct methods for promoting spheroid formation in culture, including hanging drop, liquid overlay, pellet culture, spinner culture, microfluidics, and methods incorporating biomaterial scaffolds1,17,20.
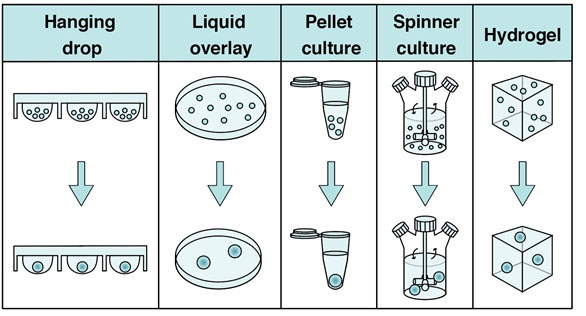
Hanging drop
One of the earliest recorded examples of the use of the hanging drop method for creating cell aggregates was in 19074. For this method, a drop of a cell suspension with a defined cell density is placed in a culture dish or plate, and the plate is flipped upside down22. The droplet remains in contact with the plate due to surface tension, while gravity forces the cells to the bottom of the droplet, allowing them to aggregate into spheroids. The size of the spheroids can be tuned by altering the droplet volume and the initial cell density.
The low cost and simplicity of this method makes it particularly amenable to high-throughput spheroid formation. As a result, robot-assisted hanging drop culturing systems using 384-well plates have been developed for drug testing purposes23.
Liquid overlay
In this case, the bottom of a cell culture dish is coated with a non-adhesive material, such as agarose, to inhibit cell adhesion and spreading. Suspension cells are then applied in a layer of media over the non-adhesive material, which drives their spontaneous aggregation and spheroid formation24. While this technique is advantageous for certain cell types, the formation of tumor spheroids is inhibited by agarose. Alternative materials can be used to promote tumor spheroid development, such as hyaluronic acid, which drives signal transduction related to cell proliferation, angiogenesis, and survival25.
Pellet culture
In this method, suspension cells are pelleted to the bottom of a tube through centrifugation, leading to cell aggregation as the cells are forced into contact. The pelleted cells are then resuspended in spheroid-promoting culture medium and placed in a plate with a non-adhesive surface coating. Pellet culture is particularly suited to the study of chondrogenesis. The pelleting of mesenchymal stem cells in this technique changes their morphology from fibroblastic to polygonal, mimicking the shape of chondrocytes and driving their differentiation along this lineage14.
Spinner culture
Suspension cells cultured in a flask with constant stirring will also form spheroids. The sizes of the resultant spheroids are partially dependent on the size of the container. The use of spinner cultures in large bioreactors has been proposed as a means of mass producing spheroids26.
Biomaterials
Spheroids can also be formed using scaffolds made from biocompatible materials like alginate, collagen, and hyaluronic acid. Hydrogels, biofilms, and particles have all been shown to improve the viability and biological properties of spheroids17. For example, hydrogels can provide an extracellular matrix-like environment with defined stiffness that can influence the malignancy of hepatocellular carcinoma cell spheroids27.
Culture conditions
The size, viability, and biological characteristics of spheroids are strongly influenced by their culture conditions. These include cell seeding number, oxygenation, media supplementation, the physicochemical properties of the culture substrates, and the inclusion of biomaterial scaffolds within the spheroids2. The optimal culture conditions for a given cell line or multicellular co-culture must be determined empirically.
The number of cells initially seeded for spheroid formation does not necessarily correlate with the size of the resultant spheroids21. In addition, spheroid formation time varies significantly and is influenced by the length of the division cycle for a given cell type. For example, while A549 adenocarcinomic lung epithelial cells can produce spheroids with the ideal morphology in 24 hours, PC-3 prostate cancer cells require around nine days in culture21.
In addition, many cell lines require medium supplemented with an extracellular matrix component. MDA-MB-231 breast cancer cells, for example, require collagen I for optimal spheroid formation21. In other cases, unique medium supplementation may be required, such as in experiments focusing on the influence of hormones and growth factors. For example, MCF7 breast cancer cell spheroids can be grown in serum-free medium supplemented with basic fibroblast growth factor to prevent unwanted estrogen receptor signaling10.
An example protocol for tumor spheroid formation
A typical protocol for tumor spheroid formation using the liquid overlay technique begins with dissociation of adherent cancer cells using trypsin21. The cells are then diluted in medium, counted, and diluted at an appropriate ratio to seed around 5,000 cells per well in a 96-well plate21. A number of plates with non-adherent coatings are commercially available21. After the cells are seeded, the plates are centrifuged at low speed for 5 – 10 minutes and then incubated under appropriate culturing conditions21. The medium is carefully changed without disturbing the spheroids, and the plates are incubated until the spheroids reach the appropriate size and/or appearance21.
Applications of spheroid culture
Tumor spheroids
A major challenge in cancer drug discovery is developing a model culture system that can faithfully recapitulate the tumor microenvironment, including the barriers to tumor penetration that exist in vivo7. The tumor microenvironment is highly complex, consisting of layers of multiple cell types, extracellular matrix components, and a hypoxic core, which cannot easily be recreated in 2D culture systems7. Indeed, many 2D cancer cell models exhibit drug sensitivity where their 3D spheroid counterparts remain resistant. For example, a synthetic tumor spheroid composed of adenocarcinomic lung epithelial cells (A549), pulmonary vascular endothelial cells, and mesenchymal stem cells resisted paclitaxel and gemcitabine treatment more so than a standard 2D co-culture of these cells29. Multicellular tumor spheroid systems like these allow researchers to evaluate the effect of drugs on tumor growth, drug penetration into tumors, cell type-specific targeting within tumors, and metastasis7.
Tissue engineering and repair
The high-throughput production of spheroids with tunable properties has been proposed as a means of engineering tissues for regenerative purposes1. Indeed, multicellular co-culture spheroids have been created which closely mimic native tissue microenvironments, including pancreatic tissue30, cardiac muscle31, the optic cup32, and cortical tissue33.
Basic biology research
Owing to their ability to mimic in vivo tissues, spheroids have been used to study a number of fundamental biological processes involving cell-cell and cell-extracellular matrix adhesion1. For example, mesenchymal stem cell spheroids were used to identify Rac1 and Cdc42 as positive regulators of chondrogenesis34.
Conclusion
In summary, spheroids are a revolutionary means of culturing cells in vitro that, by virtue of their complex 3D morphology, closely mimic the properties of their in vivo tissue counterparts. Spheroids are providing novel insights into basic biological processes, allowing for more physiologically relevant drug screening, and setting the foundations for an exciting future in tissue engineering. The low cost, potential for high-throughput culture, and ease of analysis make spheroids a great choice for many experimental applications.
Using label-free brightfield or phase contrast microscopy, healthy spheroids can be easily identified by their well-defined spherical morphology, translucent exterior and slightly darkened core, and homogeneous size. 3D cell culture techniques, including spheroid culture, will likely further gain traction in various fields as the importance of faithfully recapitulating native tissues in in vitro experiments continues to gain recognition.
References
1. Fennema, E., Rivron, N., Rouwkema, J., van Blitterswijk, C. & de Boer, J. Spheroid culture as a tool for creating 3D complex tissues. Trends Biotechnol. 31, 108–115 (2013).
2. Laschke, M. W. & Menger, M. D. Life is 3D: Boosting Spheroid Function for Tissue Engineering. Trends Biotechnol. 35, 133–144 (2017).
3. Moscona, A. THE DEVELOPMENT IN VITRO OF CHIMERIC AGGREGATES OF DISSOCIATED EMBRYONIC CHICK AND MOUSE CELLS. Proc Natl Acad Sci U S A 43, 184–194 (1957).
4. Harrison, R. G., Greenman, M. J., Mall, F. P. & Jackson, C. M. Observations of the living developing nerve fiber. Anat. Rec. 1, 116–128 (1907).
5. Sutherland, R. M., McCredie, J. A. & Inch, W. R. Growth of multicell spheroids in tissue culture as a model of nodular carcinomas. J. Natl. Cancer Inst. 46, 113–120 (1971).
6. Bissell, M. J., Hall, H. G. & Parry, G. How does the extracellular matrix direct gene expression? J. Theor. Biol. 99, 31–68 (1982).
7. Huang, B.-W. & Gao, J.-Q. Application of 3D cultured multicellular spheroid tumor models in tumor-targeted drug delivery system research. J Control Release 270, 246–259 (2018).
8. Friedrich, J., Seidel, C., Ebner, R. & Kunz-Schughart, L. A. Spheroid-based drug screen: considerations and practical approach. Nat Protoc 4, 309–324 (2009).
9. Zhang, K., Yan, S., Li, G., Cui, L. & Yin, J. In-situ birth of MSCs multicellular spheroids in poly(L-glutamic acid)/chitosan scaffold for hyaline-like cartilage regeneration. Biomaterials 71, 24–34 (2015).
10. Generation of MCF7 spheroids in serum-free conditions. (2019). at https://assets.thermofisher.co...;
11. Jo, Y. H. et al. Artificial islets from hybrid spheroids of three pancreatic cell lines. Transplant. Proc. 46, 1156–1160 (2014).
12. Antoni, D., Burckel, H., Josset, E. & Noel, G. Three-dimensional cell culture: a breakthrough in vivo. Int J Mol Sci 16, 5517–5527 (2015).
13. Yoon, H. H., Bhang, S. H., Shin, J.-Y., Shin, J. & Kim, B.-S. Enhanced cartilage formation via three-dimensional cell engineering of human adipose-derived stem cells. Tissue Eng Part A 18, 1949–1956 (2012).
14. Zhang, L. et al. Chondrogenic differentiation of human mesenchymal stem cells: a comparison between micromass and pellet culture systems. Biotechnol. Lett. 32, 1339–1346 (2010).
15. Groebe, K. & Mueller-Klieser, W. On the relation between size of necrosis and diameter of tumor spheroids. Int. J. Radiat. Oncol. Biol. Phys. 34, 395–401 (1996).
16. Bhang, S. H. et al. Angiogenesis in ischemic tissue produced by spheroid grafting of human adipose-derived stromal cells. Biomaterials 32, 2734–2747 (2011).
17. Ryu, N.-E., Lee, S.-H. & Park, H. Spheroid Culture System Methods and Applications for Mesenchymal Stem Cells. Cells 8, 1620 (2019).
18. Lin, R.-Z., Chou, L.-F., Chien, C.-C. M. & Chang, H.-Y. Dynamic analysis of hepatoma spheroid formation: roles of E-cadherin and beta1-integrin. Cell Tissue Res. 324, 411–422 (2006).
19. Tsai, A.-C., Liu, Y., Yuan, X. & Ma, T. Compaction, fusion, and functional activation of three-dimensional human mesenchymal stem cell aggregate. Tissue Eng Part A 21, 1705–1719 (2015).
20. Torisawa, Y.-S. et al. Efficient formation of uniform-sized embryoid bodies using a compartmentalized microchannel device. Lab Chip 7, 770–776 (2007).
21. Generation of cancer spheroids— tips and tricks. (2020). doi:10.1083/jcb.105.1.117
22. Bartosh, T. J. & Ylostalo, J. H. Preparation of anti-inflammatory mesenchymal stem/precursor cells (MSCs) through sphere formation using hanging-drop culture technique. Curr Protoc Stem Cell Biol 28, Unit 2B.6.–2B.6.23 (2014).
23. Tung, Y.-C. et al. High-throughput 3D spheroid culture and drug testing using a 384 hanging drop array. Analyst 136, 473–478 (2011).
24. Costa, E. C., de Melo-Diogo, D., Moreira, A. F., Carvalho, M. P. & Correia, I. J. Spheroids Formation on Non-Adhesive Surfaces by Liquid Overlay Technique: Considerations and Practical Approaches. Biotechnol J 13, 1700417 (2018).
25. Carvalho, M. P., Costa, E. C., Miguel, S. P. & Correia, I. J. Tumor spheroid assembly on hyaluronic acid-based structures: A review. Carbohydr Polym 150, 139–148 (2016).
26. Want, A. J., Nienow, A. W., Hewitt, C. J. & Coopman, K. Large-scale expansion and exploitation of pluripotent stem cells for regenerative medicine purposes: beyond the T flask. Regen Med 7, 71–84 (2012).
27. Liang, Y. et al. A cell-instructive hydrogel to regulate malignancy of 3D tumor spheroids with matrix rigidity. Biomaterials 32, 9308–9315 (2011).
28. Vinci, M. et al. Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol. 10, 29–21 (2012).
29. Lamichhane, S. P. et al. Recapitulating epithelial tumor microenvironment in vitro using three dimensional tri-culture of human epithelial, endothelial, and mesenchymal cells. BMC Cancer 16, 581–12 (2016).
30. Lumelsky, N. et al. Differentiation of embryonic stem cells to insulin-secreting structures similar to pancreatic islets. Science 292, 1389–1394 (2001).
31. Kehat, I. et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J. Clin. Invest. 108, 407–414 (2001).
32. Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011).
33. Eiraku, M. et al. Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 3, 519–532 (2008).
34. Woods, A., Wang, G., Dupuis, H., Shao, Z. & Beier, F. Rac1 signaling stimulates N-cadherin expression, mesenchymal condensation, and chondrogenesis. J. Biol. Chem. 282, 23500–23508 (2007).

