Study live cells using non-invasive & non-toxic methods
Live-cell imaging has become a fundamental tool to better understand cellular processes and biological functions. Cells are, however, as translucent as ice cubes in water. As a result, fluorescent labeling techniques are commonly used to enhance brightfield images, to visualize proteins and subcellular compartments to help us answer biological questions and make new discoveries.
While labeling techniques are powerful visualization tools, the labeling process is invasive and the lasers used in fluorescence imaging can be toxic to cells. This can cause abnormal cellular behavior and compromise research findings.
A non-invasive and non-toxic alternative to fluorescent microscopy is label-free imaging. Here, a combination of contrast-improving optics and image analysis algorithms are used to analyze cell cultures. Although the resolution achievable through label-free imaging does not typically provide the level of detail that is seen in fluorescent imaging, label-free imaging can provide a wealth of valuable biological information through the analysis of cellular dynamics and morphology.
This blog touches on the drawbacks associated with live-cell fluorescence imaging and details the dynamic and morphological information that can be obtained through label-free imaging.
------------------------------------------------------
Researchers can perform various experiments with these CytoSMART label-free live-cell imagers: full-plate scanner Omni, mini live-cell imagers Lux2 and Lux3 BR.
------------------------------------------------------
Fluorescence microscopy
Fluorescence microscopy uses fluorescent dyes (fluorophores), which are molecules that absorb one high(er) energy wavelength of light (excitation) and emit another low(er) energy wavelength (emission). Live-cell staining of cellular organelles, such as the nucleus, cytoplasm, cytoskeleton, plasma membranes, ER, Golgi, lysosomes, and mitochondria is possible through the use of these fluorescent dyes. These dyes bind to organelles based on their characteristics, such as membrane potential, pH, and lipophilicity. While using fluorescent dyes is relatively straightforward, these dyes have varying levels of toxicity towards cells1.
To track specific proteins using live-cell fluorescence imaging, tags such as fluorescent proteins or peptides need to be genetically introduced into cells. This involves fusing a fluorescent protein (such as the well-known green fluorescent protein, GFP) to a target protein of interest, which is then either introduced into the genome of the cell or expressed from a plasmid2.
These genetic modifications can trigger abnormal cell behavior. For example, the location of genome integration could interfere with normal cell functioning; another possibility is that high levels of ectopic expression of a fluorescently labeled target could alter behavior or even become toxic to cells1. Furthermore, fluorescent proteins are large and can hinder the normal functioning of the protein of interest (this is less of an issue with fluorescent peptides which are much smaller), which can alter normal behavior and/or mislead your findings3.
A major drawback of live fluorescence imaging is phototoxicity, which cal be recognized by looking for phenotypic indications (Figure 1). The low- and high-wavelength light emitted by lasers to excite fluorophores becomes toxic to cells. Furthermore, photoexcited fluorophores can release reactive oxygen species, which damage cellular structures. These perturbations cause cells to behave abnormally and/or die. This interferes with the experimental process and greatly limits the time live cells can be analyzed under a fluorescent microscope1,3.
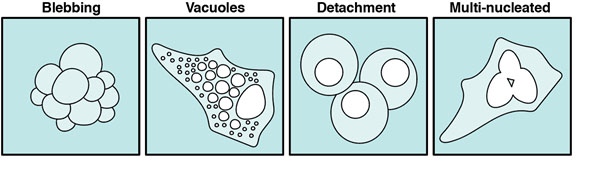
Figure 1 | Phenotypes indicative of cells stress or apoptosis.
What is label-free imaging?
Label-free imaging, as the name suggests, can be defined as a method for visualizing cells that have not been labeled or altered in any way. To achieve this goal brightfield, phase contrast, and differential interference contrast microscopy can be used to visualize label-free cells. The two latter techniques are used to improve on the image quality of standard brightfield microscopy.
Most cells are thin and transparent; they do not absorb much light and are therefore difficult to see without adding optics that allow the phase shift of light induced by the sample (Figure 2). The two most commonly used techniques to visualize this phase shift are phase contrast, which causes cells to appear dark on a light background, and differential interference contrast, which gives a pseudo–3D shaded appearance to cells (Figure 2)2.
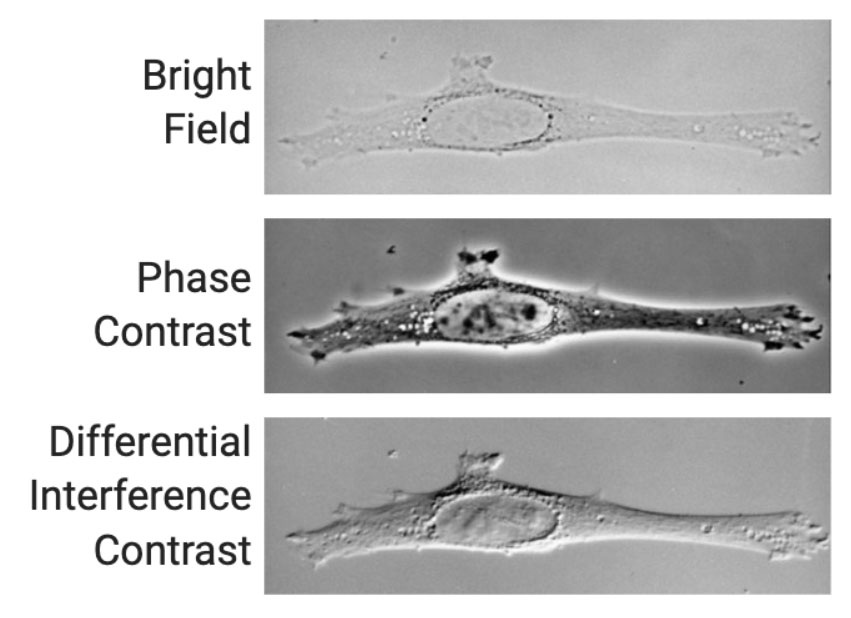
Although the applications for label-free imaging are not as extensive as fluorescence imaging, they do however have several obvious benefits.
Firstly and most importantly, cells are being analyzed in their native, unperturbed state. Findings are therefore more reliable and biologically relevant. Secondly, it is a much quicker and cheaper technique as there is no genetic modification and cell line generation (which is extremely time-consuming and often requires multiple attempts before getting it right), no fluorescence dyes or tags to purchase (these are costly) and complex lasers to run.
Another advantage is that the duration of time-lapse experiments is not limited by the threat of phototoxicity. So in theory, if cells are kept in a tightly controlled environment (i.e. appropriate temperature, CO2 levels, pH) with sufficient nutrient supplies, experiments could go on for weeks or even months depending on your experimental context.
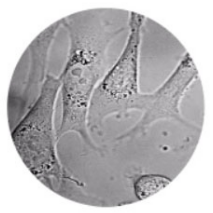
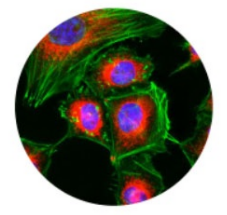
Table 3 | A comparison between label-free and fluorescence live-cell imaging techniques.
| Label-free | Fluorescence | |
|---|---|---|
 |
 |
|
| Applications | Study cellular dynamics & cell morphology. | Study cellular processes and functions. Visualise specific structures and proteins. |
| Time | Quick | Can be very time consuming |
| Cost | Cheap | Can be very expensive. |
| Invasiveness | Non-invasive. Cells unperturbed and in their natural state. | Invasive labelling-techniques and phototoxicity |
| Measurement periods | Can analyze cells over long periods of time. | Shorter periods of analysis due to phototoxicity. |
ApplicationsStudy cellular dynamics & cell morphology.Study cellular processes and functions. Visualise specific structures and proteins.
Label-free metrics and their biological relevance
Cells can be highly dynamic with changes in their morphology and behavior. Live-cell label-free imaging enables insight into these changes over time.
Label-free imaging enables the identification and quantification of cellular events such as cell division, proliferation, motility, migration, differentiation, and death (Figure 4). These are fundamental processes in development, tissue repair, and immune regulation. Importantly, deregulation of these processes can result in diseases such as cancer, autoimmune disorders, neurological disease, and chronic inflammation. Therefore, label-free imaging has huge potential in better understanding and characterizing cell behavior in the context of these processes under different experimental conditions4.
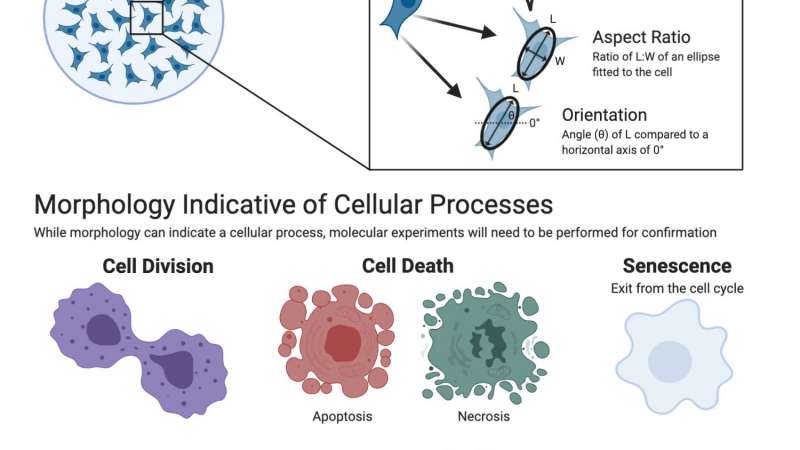
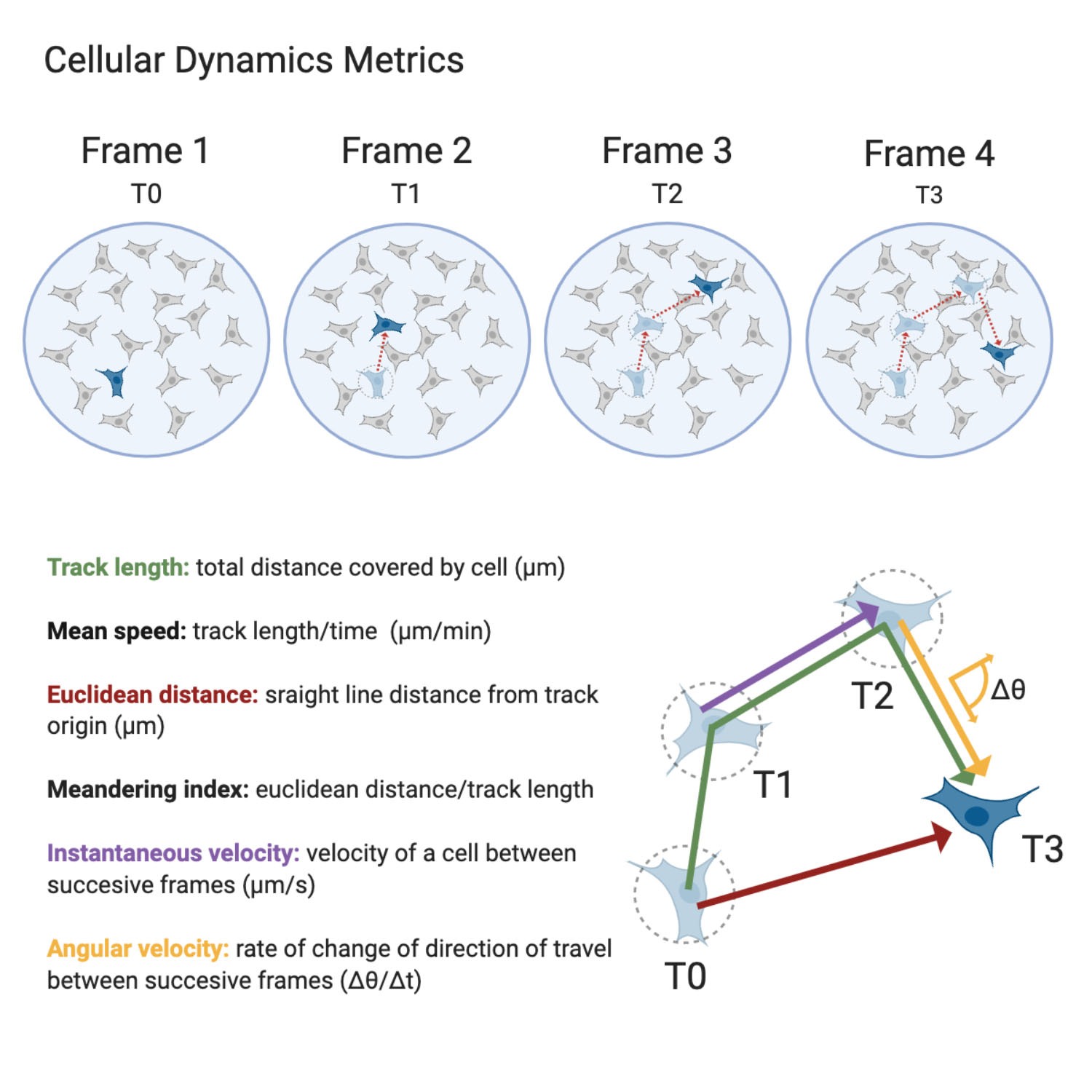
Furthermore, the ability to track single-cell behavior and morphology over time (not population averages) is a powerful tool to gain insight into cell-to-cell variability within a population. This has implications for understanding responses to treatments or stimuli and therapeutic resistance. Figure 4 and Figure 5 highlight the morphology and dynamic behavior metrics, respectively, that can be extracted from single-cells using label-free imaging4,5,6.
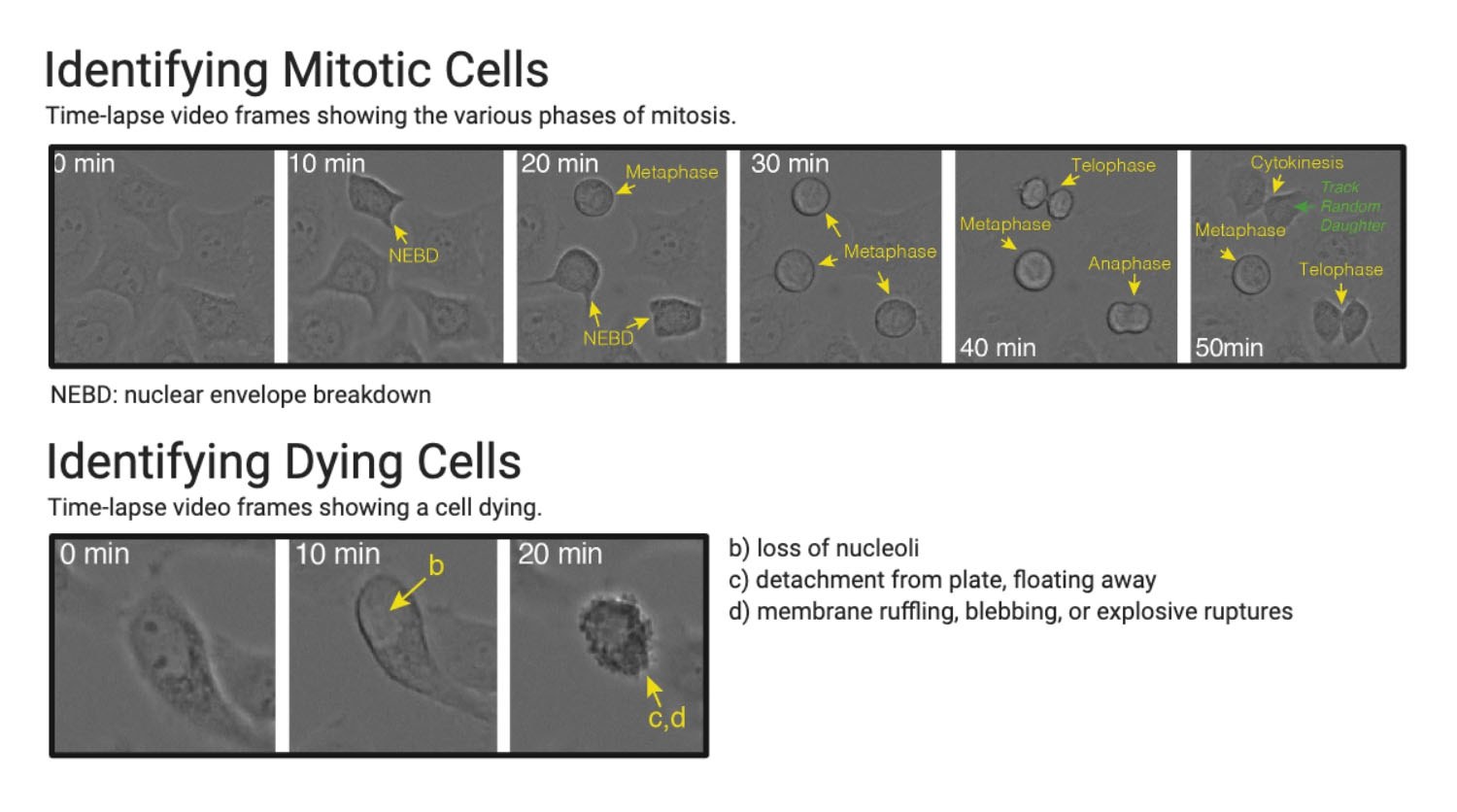
A recent study conducted by Caldon and Burgess5 is a good example of label-free morphology metrics being used to track the fate of single-cells in brightfield (using 10-20X magnification) time-lapse videos. The authors were able to successfully track and quantify cell cycle length, cell fate, and mitotic phenotypes (the protocol and software used are all open-source). The images in Figure 6 show examples of the morphological features that they were able to identify and quantify from their time-lapse video. A study by Kasprowicz et al. is an example of measuring cellular dynamics metrics to characterize single-cell cell behavior. This study highlights the power of tracking single-cells to analyze behavior change compared to population averages6.
Concluding remarks
Live fluorescence microscopy has contributed significantly to our understanding of cell structure and function, processes and interactions and is a fundamental field in cell biology that will continue to advance. While there are drawbacks with using fluorescence dyes and tags, there are numerous experimental controls that can be used to help identify any confounding effects, but phototoxicity still remains a challenge for longer periods of imaging.
Label-free imaging, however, is an economical and quick technique with untapped potential. The ability to fate-track single cells (for as long as you want) and quantify morphological and cellular dynamics in an unperturbed, natural environment will be critically important to study specific events such as drug response, therapeutic resistance, and differentiation.
Related Products
There are currently no products tagged to this resource.